Abstracts of the 13th International Conference on Thrombosis and Hemostasis Issues in Cancer, 2026
Vol. 5 No. s1 (2026)
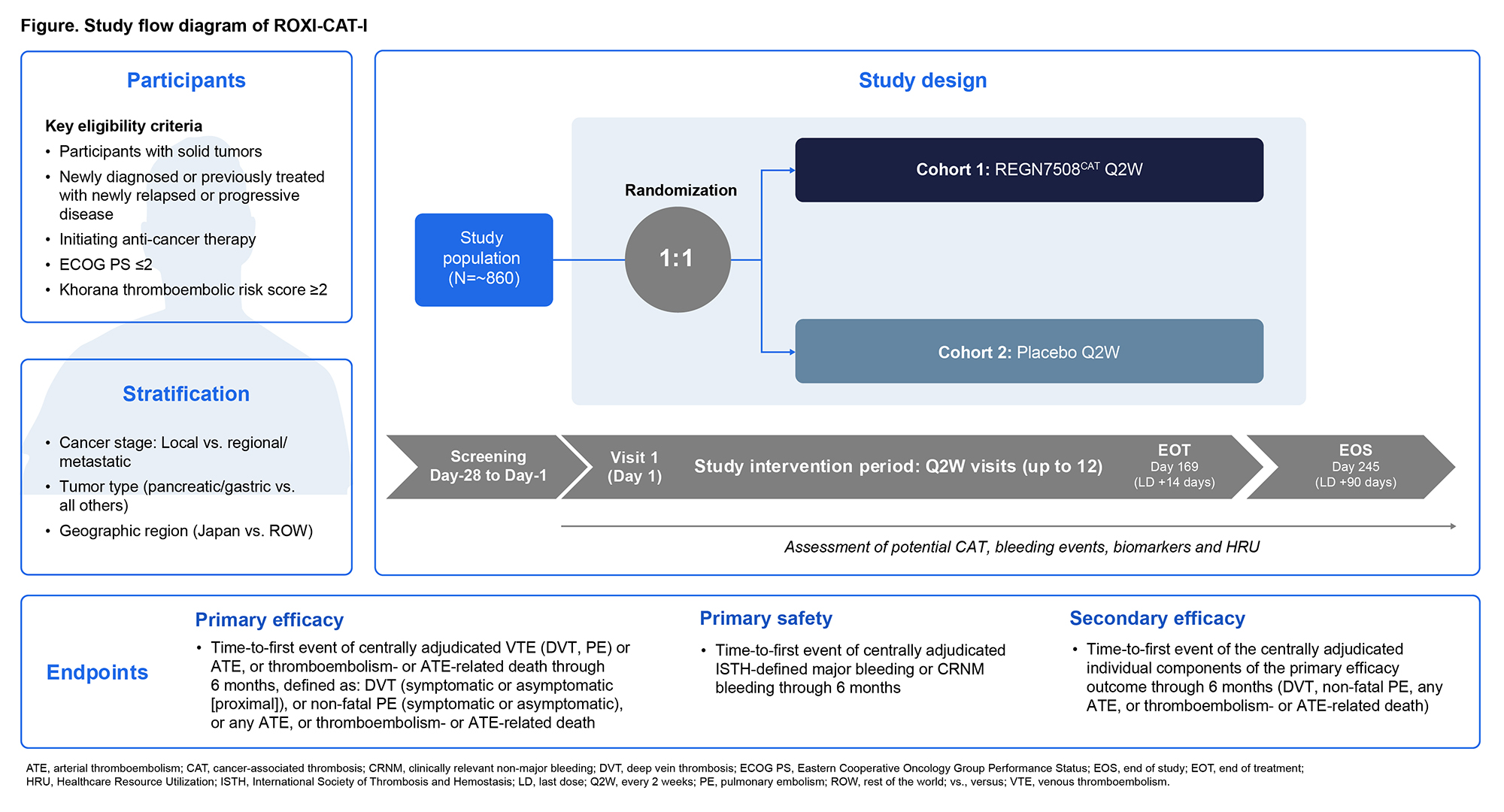
PO53 | PHASE 3 TRIAL EVALUATING THE EFFICACY AND SAFETY OF REGN7508CAT FOR PRIMARY PROPHYLAXIS OF CANCER-ASSOCIATED THROMBOSIS FOR PARTICIPANTS WITH SOLID TUMORS UNDERGOING CANCER TREATMENT (ROXI-CAT-I)
J. Zwicker1, M. P. O’Brien2, M. Carrier3, J. Connors4, A. Falanga5, A. Manzano6, R. Mcbane7, Y.Y. Janjigian1, G. O’Kane8, M. Onisko2, J. Kaplan2, A.P. Kithcart2, D.E. Gutstein2, A. Khorana9, M. F. Walsh2 | 1Memorial Sloan Kettering Cancer Center, New York, USA; 2Regeneron Pharmaceuticals, Inc. , Tarrytown, NY, USA; 3University of Ottawa, Ottawa, ON, Canada; 4Harvard Medical School, Boston, MA, USA; 5Hospital Papa Giovanni XXIII, Bergamo, Italy; 6Hospital Universitario 12 de Octubre, Madrid, Spain; 7Mayo Clinic, Rochester, MN, USA; 8St. Vincent's University Hospital and School of Medicine, University College Dublin, Dublin, Ireland; 9James P. Wilmot Cancer Center and the Department of Medicine, University of Rochester, Rochester, NY, USA

Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 16 April 2026
2
Views
0
Downloads
Most read articles by the same author(s)
- Società Italiana di Emostasi e Trombosi, PO70 | HEMOSTATIC ABNORMALITIES IN A PATIENT WITH WALDENSTRÖM'S MACROGLOBULINEMIA , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO38 | PERICATHETER AND SYSTEMIC THROMBOSIS DURING CAR-T LYMPHOCYTAPHERESIS: INCIDENCE AND CLINICAL CORRELATES , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO44 | PREDICTORS OF ONE-YEAR MORTALITY IN NEWLY DIAGNOSED METASTATIC BREAST CANCER: THE PROGNOSTIC ROLE OF HEMOSTATIC BIOMARKERS AND EARLY THROMBOEMBOLISM , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)