Abstracts of the 13th International Conference on Thrombosis and Hemostasis Issues in Cancer, 2026
Vol. 5 No. s1 (2026)
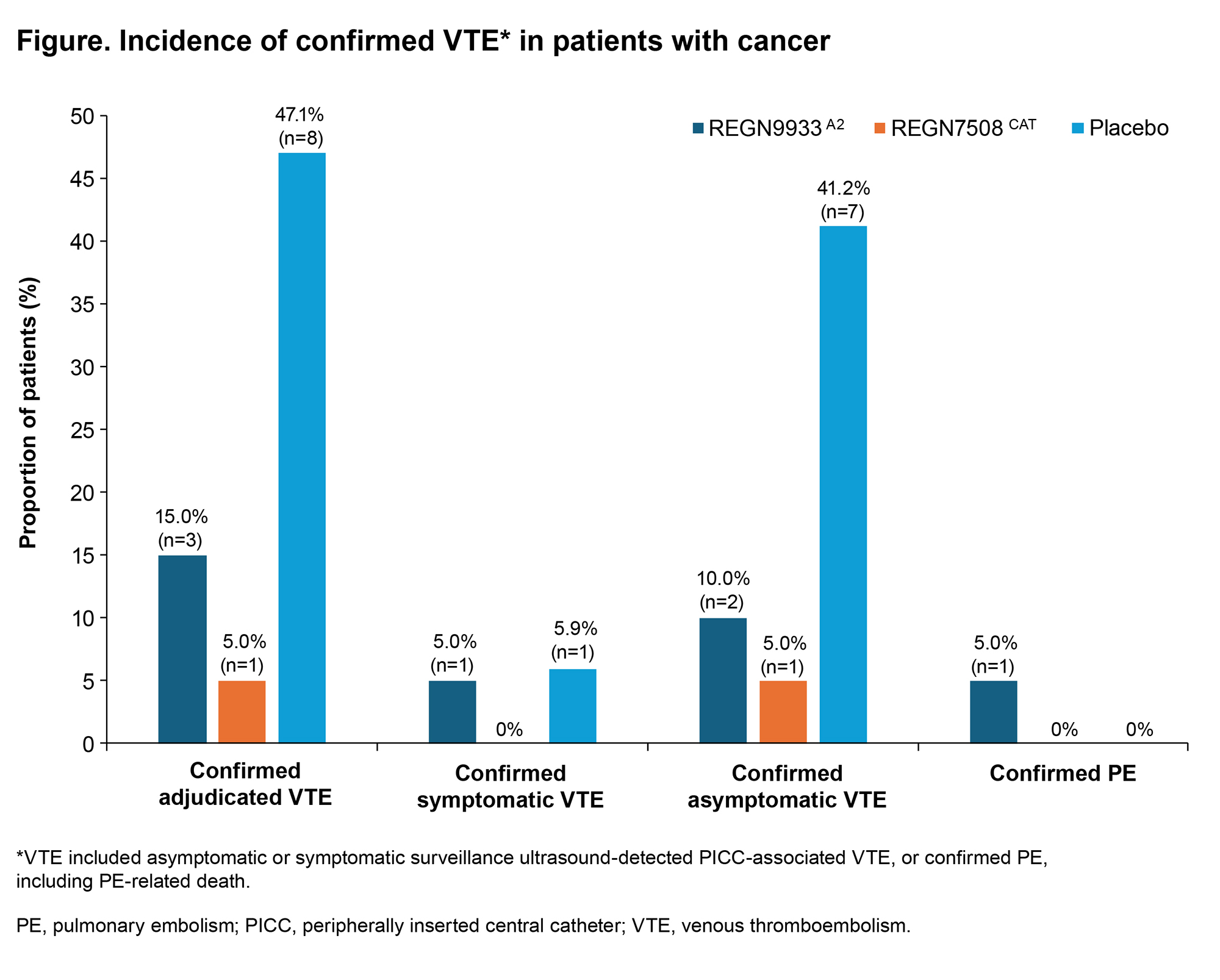
PO50 | SUB-ANALYSIS OF INTERIM RESULTS FROM A PHASE 2 STUDY INVESTIGATING REGN9933A2 AND REGN7508CAT FOR THE PREVENTION OF CONTACT-MEDIATED VENOUS THROMBOEMBOLISM IN PATIENTS WITH ACTIVE CANCER UNDERGOING PERIPHERALLY INSERTED CENTRAL CATHETER PLACEMENT (ROXI-CATH)
J. Zwicker1,2, A.P. Kithcart3, J. Kaplan3, Y-C. Cheng3, J.G. Raya3, J. Xiao3, S. Li3, K. Musgrave4, D.E. Gutstein3, G. Piazza5 | 1Memorial Sloan Kettering Cancer Center, New York, NY, USA; 2Weill Cornell Medical Center, New York, NY, USA; 3Regeneron Pharmaceuticals, Inc. , Tarrytown, NY, USA; 4Newcastle upon Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, UK; 5Brigham and Women’s Hospital, Boston, MA, USA

Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 16 April 2026
1
Views
0
Downloads
Most read articles by the same author(s)
- Società Italiana di Emostasi e Trombosi, PO04 | FIBRIN CLOT SHIELDS PROMOTE CLONAL SELECTION IN CANCER CELLS: PROCOAGULANT ACTIVITY AND SURVIVAL OF CLOT-EMBEDDED CELLS. A NOVEL TUMOR MICROENVIRONMENT MODEL , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO12 | IMPACT OF INHERITED THROMBOPHILIA ON CANCER-ASSOCIATED THROMBOSIS: A SYSTEMATIC REVIEW AND META-ANALYSIS , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO29 | CANADIAN INSIGHTS INTO THE MANAGEMENT OF BREAKTHROUGH THROMBOSIS , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO51 | MECHANISTIC POPULATION PHARMACOKINETIC/PHARMACODYNAMIC MODELING OF FACTOR XI-TARGETING MONOCLONAL ANTIBODIES SUPPORTS DURABLE ANTICOAGULANT COVERAGE AND TRANSLATIONAL DOSE AND REGIMEN SELECTION , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, OC10 | FROM DESIGN TO CLINICAL PHASE 3 IN ONCOLOGY: CD13-TARGETED TISSUE FACTOR AND TUMOR INFARCTION , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO05 | DESIGN, SYNTHESIS, AND BIOLOGICAL EVALUATION OF NOVEL IMATINIB AND NILOTINIB ANALOGUES EXPRESSING ENHANCED ANTIPLATELET AND ANTICANCER ACTIVITIES , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO22 | LONG-TERM ANTICOAGULANT THERAPY AND REDUCTION OF OVARIAN CANCER RECURRENCE: CLINICAL EVIDENCE OF A POTENTIAL ANTITUMOUR EFFECT , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO06 | ACTIVATED PLATELETS INDUCE THE FORMATION OF NEUTROPHIL EXTRACELLULAR TRAPS VIA TOLL-LIKE RECEPTOR-4 AND P-SELECTIN GLYCOPROTEIN LIGAND-1 , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO53 | PHASE 3 TRIAL EVALUATING THE EFFICACY AND SAFETY OF REGN7508CAT FOR PRIMARY PROPHYLAXIS OF CANCER-ASSOCIATED THROMBOSIS FOR PARTICIPANTS WITH SOLID TUMORS UNDERGOING CANCER TREATMENT (ROXI-CAT-I) , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO62 | CLINICAL AND MOLECULAR DIAGNOSTIC CHALLENGES IN MYELOPROLIFERATIVE NEOPLASMS WITH LOW JAK2V617F ALLELIC BURDEN , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)