Abstracts of the 13th International Conference on Thrombosis and Hemostasis Issues in Cancer, 2026
Vol. 5 No. s1 (2026)
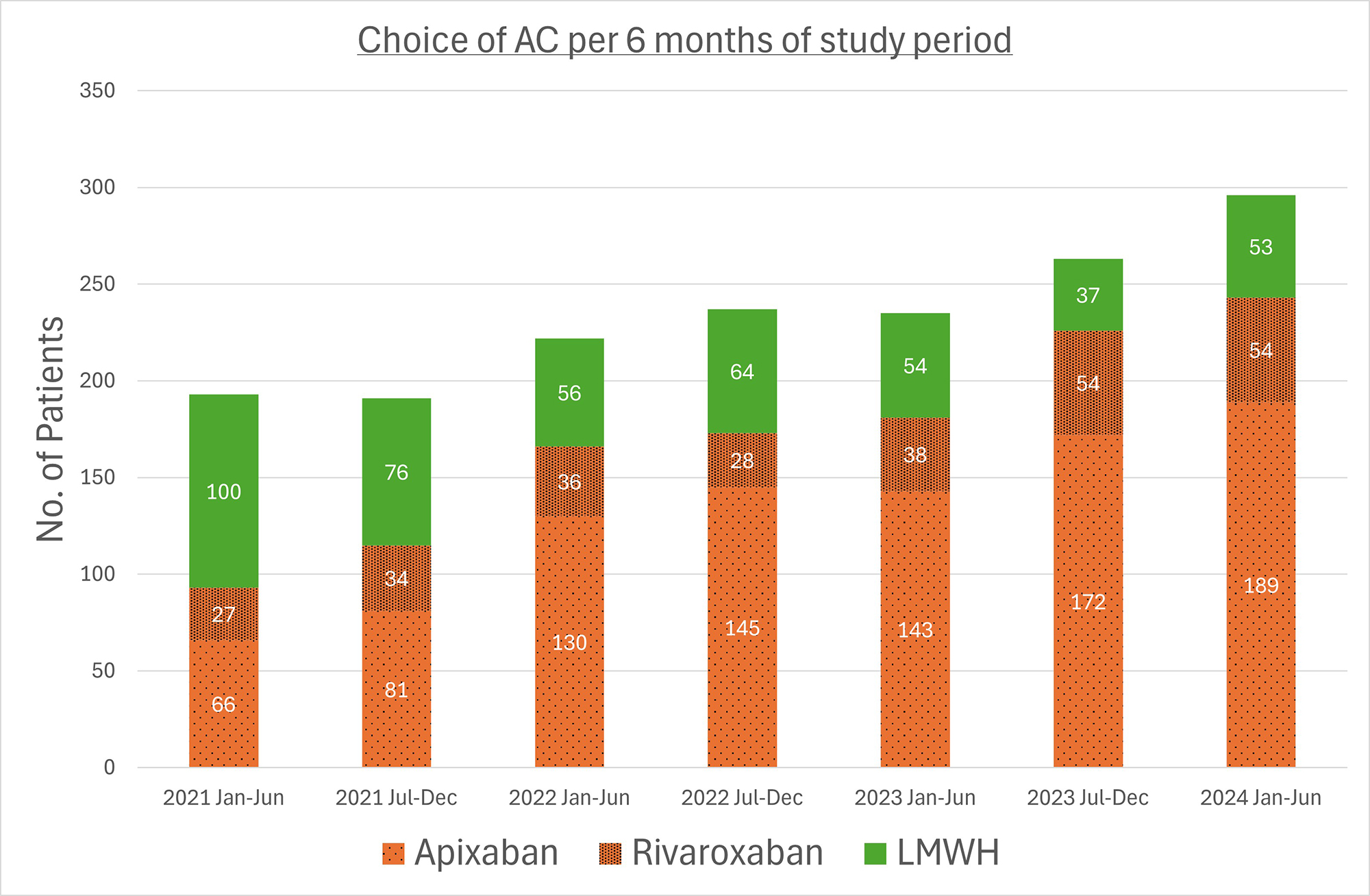
OC04 | DIRECT ORAL ANTICOAGULANTS PROVIDE EFFECTIVE THROMBOPROPHYLAXIS IN NEWLY DIAGNOSED MYELOMA: REAL WORLD FINDINGS FROM THE ATOMM STUDY
T. Bull1, Heamstar Collaborators2, W. Wilson3, E. Ganendra4, M. Thomas5, R. Alikhan6, M. Karanth1, M. Camilleri7 | 1West Suffolk Hospital NHS Foundation Trust, UK; 2Haematology Specialty Training Audit and Research HaemSTAR Network, UK; 3University College London Clinical Trial Centre, UK; 4North West Anglia NHS Foundation Trust, UK; 5University College London Hospitals, UK; 6Cardiff and Vale University Health Board, Wales, UK; 7Cambridge University Hospitals NHS Foundation Trust, UK

Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 16 April 2026
108
Views
28
Downloads
Most read articles by the same author(s)
- Società Italiana di Emostasi e Trombosi, PO52 | MECHANISTIC POPULATION PHARMACOKINETIC/PHARMACODYNAMIC AND TIME-TO-EVENT MODELING SUPPORT SUSTAINED FACTOR XI INHIBITION AND SUPERIOR POST-OPERATIVE VENOUS THROMBOEMBOLISM PREVENTION WITH REGN7508CAT , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO49 | PROPHYLACTIC ANTICOAGULATION DECISIONS IN HIGH-RISK PATIENTS RECEIVING CANCER-DIRECTED THERAPY: ANALYSIS OF THE VERMONT METHOD , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO02 | SYNERGISTIC INTERACTION OF ENDOTHELIAL AND CANCER CELLS IN THE FORMATION AND STRUCTURE OF THE FIBRIN CLOT SHIELDS , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO64 | MANAGEMENT OF BLEEDING RISK IN PATIENTS WITH HEPATOCELLULAR CARCINOMA RECEIVING SYSTEMIC THERAPY: A SURVEY OF CLINICAL PRACTICE , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, OC08 | A SILK-BASED 3D BONE MARROW MODEL FOR CHEMOTHERAPY-INDUCED THROMBOCYTOPENIA , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO69 | INCIDENTAL CANCER DETECTED DURING EMERGENCY DIAGNOSTIC WORK-UP FOR ACUTE PULMONARY EMBOLISM: A RETROSPECTIVE COHORT STUDY , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO42 | DIFFERENTIAL EXPRESSION OF HEMOSTATIC BIOMARKERS ACROSS CANCER TYPE AND DISEASE STAGE: A PROSPECTIVE ANALYSIS OF 4,292 PATIENTS FROM THE HYPERCAN STUDY , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO70 | HEMOSTATIC ABNORMALITIES IN A PATIENT WITH WALDENSTRÖM'S MACROGLOBULINEMIA , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO54 | CANCER-ASSOCIATED THROMBOSIS IN ASPIRIN-TREATED PATIENTS AFTER ARTERIAL THROMBOSIS: THE COMPASS-ARTECAT-ASA PROSPECTIVE COHORT STUDY , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Società Italiana di Emostasi e Trombosi, PO40 | RADAR, UK-MRA MYELOMA XV - COMPARING MRD-GUIDED TREATMENT ESCALATION AND DE-ESCALATION STRATEGIES IN PATIENTS WITH NEWLY DIAGNOSED MYELOMA SUITABLE FOR STEM CELL TRANSPLANTATION: THROMBOPROPHYLAXIS SUBGROUP STUDY PROTOCOL , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)