Unlocking the potential of statins for venous thromboembolism prophylaxis in cancer: why conduct the STAT-CAT trial?

All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
Patients with cancer have an increased risk of venous thromboembolism (VTE), contributing to excess morbidity, mortality, treatment delays, and healthcare utilization. The incidence of cancer-associated VTE has increased over the past two decades. Patients with malignancy now comprise approximately 20% of the global VTE burden. VTE risk is highest during the first 3–6 months after a new or recurrent cancer diagnosis. A new diagnosis of VTE can herald the presence of cancer. Although the risk declines over time, it may not return to baseline for up to two years after cancer treatment in those with no evidence of disease. Randomized trials of prophylactic-dose anticoagulation for primary VTE prevention in ambulatory patients with cancer, including the direct oral anticoagulants, demonstrate a 35–60% reduction in VTE events; however, routine use of primary prophylaxis remains quite limited among patients with cancer due to excessive bleeding risks, which are doubled with most anticoagulants. These competing risks are reflected in conditional guideline recommendations from major societies, which suggest consideration of prophylaxis only in selected high-risk patients with concomitantly low bleeding risks. By contrast, in the general population, statin therapy has been shown to reduce VTE rates by 30 to 40% with no increase in hemorrhage and thus might be a highly effective intervention to reduce the risk of cancer-associated thrombosis. The Statin Therapy to Prevent Cancer-Associated VTE (STAT-CAT) trial, funded by the NHLBI, has been designed to directly test this hypothesis among 4,000 patients initiating cancer therapy. This review summarizes emerging evidence supporting statins as a potential approach to primary VTE prevention in patients with cancer and reviews clinical trials that address statins for VTE risk reduction.

Graphical Abstract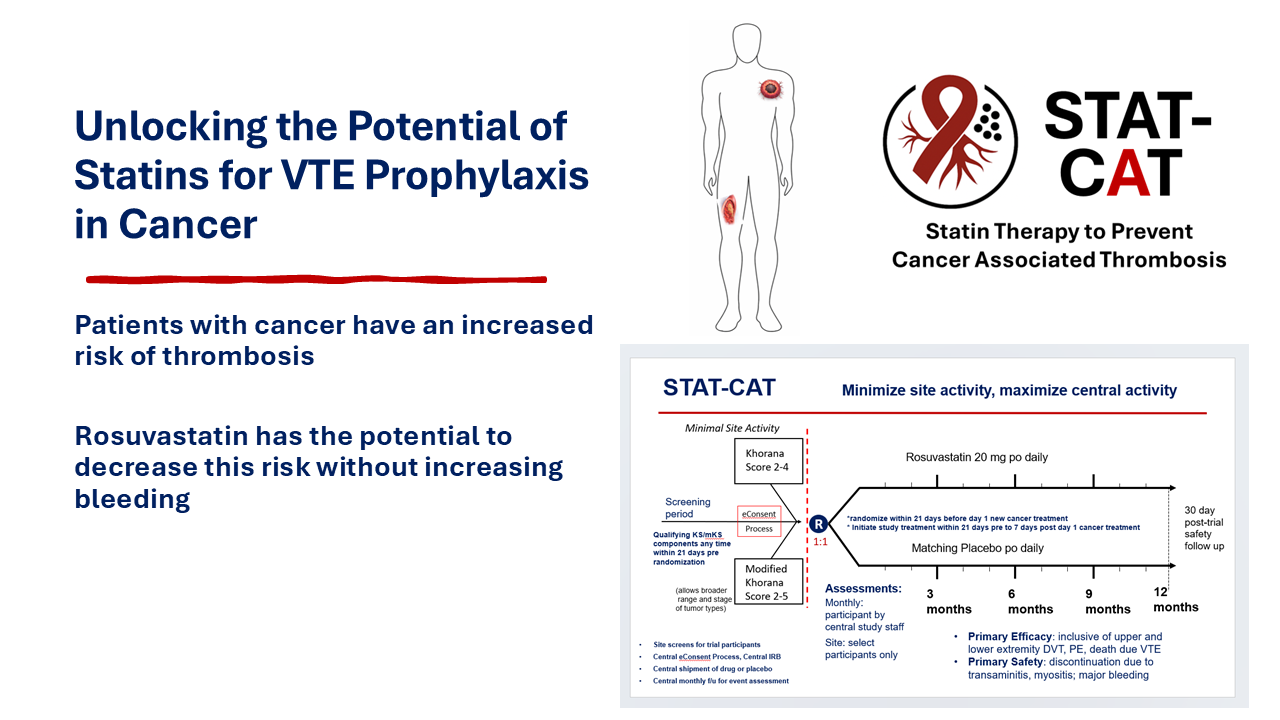
Supporting Agencies
STAT-CAT is funded through NHLBI grant UG3HL176627How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Most read articles by the same author(s)
- Anna Falanga, Benjamin Brenner, Alok A. Khorana, Welcome to the 12th International Conference on Thrombosis and Hemostasis Issues in Cancer! , Bleeding, Thrombosis and Vascular Biology: Vol. 3 No. s1 (2024)
- Mehrie H. Patel, Alok A. Khorana, New drugs, old problems: immune checkpoint inhibitors and cancer-associated thrombosis , Bleeding, Thrombosis and Vascular Biology: Vol. 3 No. s1 (2024)
- Anna Falanga, Benjamin Brenner, Alok A. Khorana, Welcome to the 12th International Conference on Thrombosis and Hemostasis Issues in Cancer! , Bleeding, Thrombosis and Vascular Biology: Vol. 3 No. s1 (2024)
- Anna Falanga, Benjamin Brenner, Alok A. Khorana, Preface to the 13th International Conference on Thrombosis and Hemostasis Issues in Cancer, 2026 , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)
- Hiranya M. Dave, Mehrie H. Patel, Keith R. McCrae, Alok A. Khorana, Factor XI inhibitors in cancer-associated venous thromboembolism: what’s next? , Bleeding, Thrombosis and Vascular Biology: Vol. 5 No. s1 (2026)









