Cancer-associated thrombosis: limitations of conventional models and advances with endothelial cell-based microfluidics incorporating thrombin generation

All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
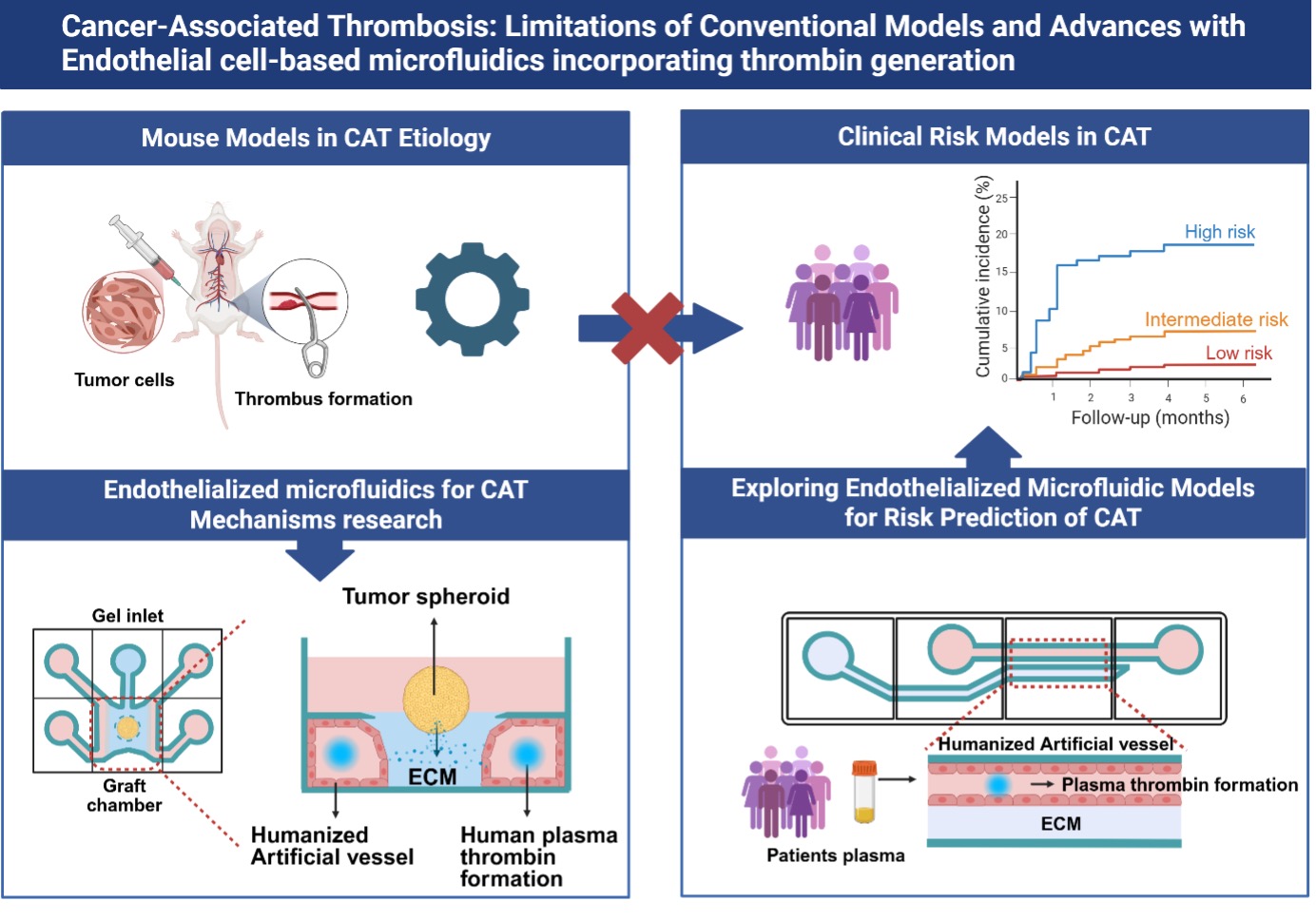
Apart from cancer progression itself, cancer-associated thrombosis (CAT) is the second leading cause of death among cancer patients. However, the mechanisms underlying CAT remain incompletely understood. Traditional animal models cannot fully reproduce the complex pathophysiological process of CAT in humans. Meanwhile, existing clinical prediction models are still insufficient to provide reliable risk prediction tools for venous thromboembolism (VTE) in cancer patients. With the accelerating development of microfluidics technology, it has become possible to construct thrombotic microenvironments with high physiological relevance in vitro, providing a new platform for the study of CAT. In this review, we summarized the contributions and limitations of animal models and clinical studies to CAT, and highlighted a series of artificial vascular microfluidics developed by our research group in recent years that can, to some extent, simulate the in vivo microenvironment for CAT. Based on this microfluidics platform, we conducted thrombin generation analysis to explore the potential mechanisms of cancer-related thrombosis and predict VTE risk in cancer patients. Finally, we further discuss the key issues and the challenges in the future.

Graphical Abstract
Supporting Agencies
China Scholarship CouncilHow to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.









